No-Touch Breast Augmentation in San Francisco: How This Technique Lowers Your Risk of Capsular Contracture
Overview
The no-touch technique keeps the breast implant completely isolated from your skin bacteria during insertion, and it reduces capsular contracture rates by up to 87%.
Capsular contracture, where scar tissue tightens around the implant, is the number-one reason patients need revision breast surgery.
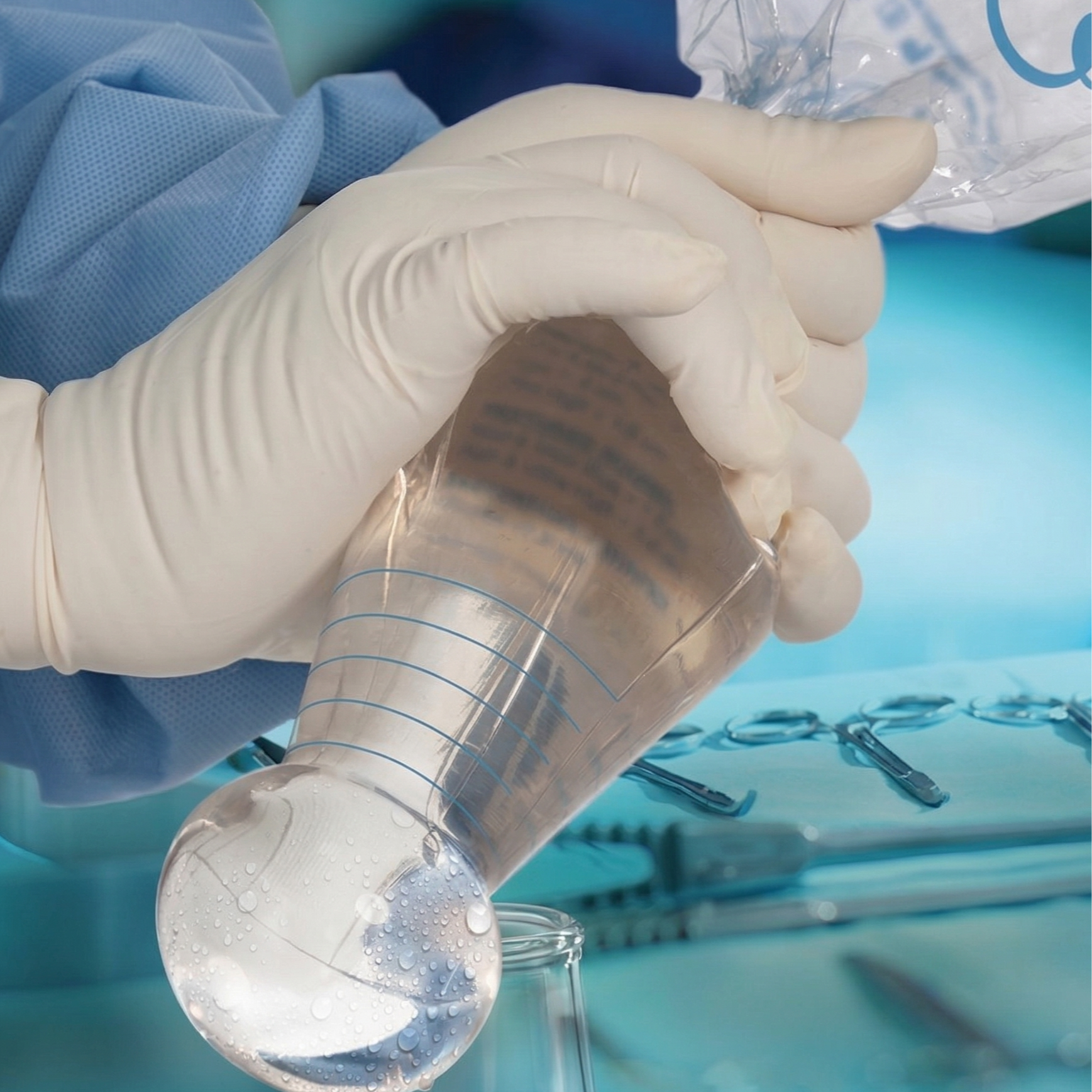
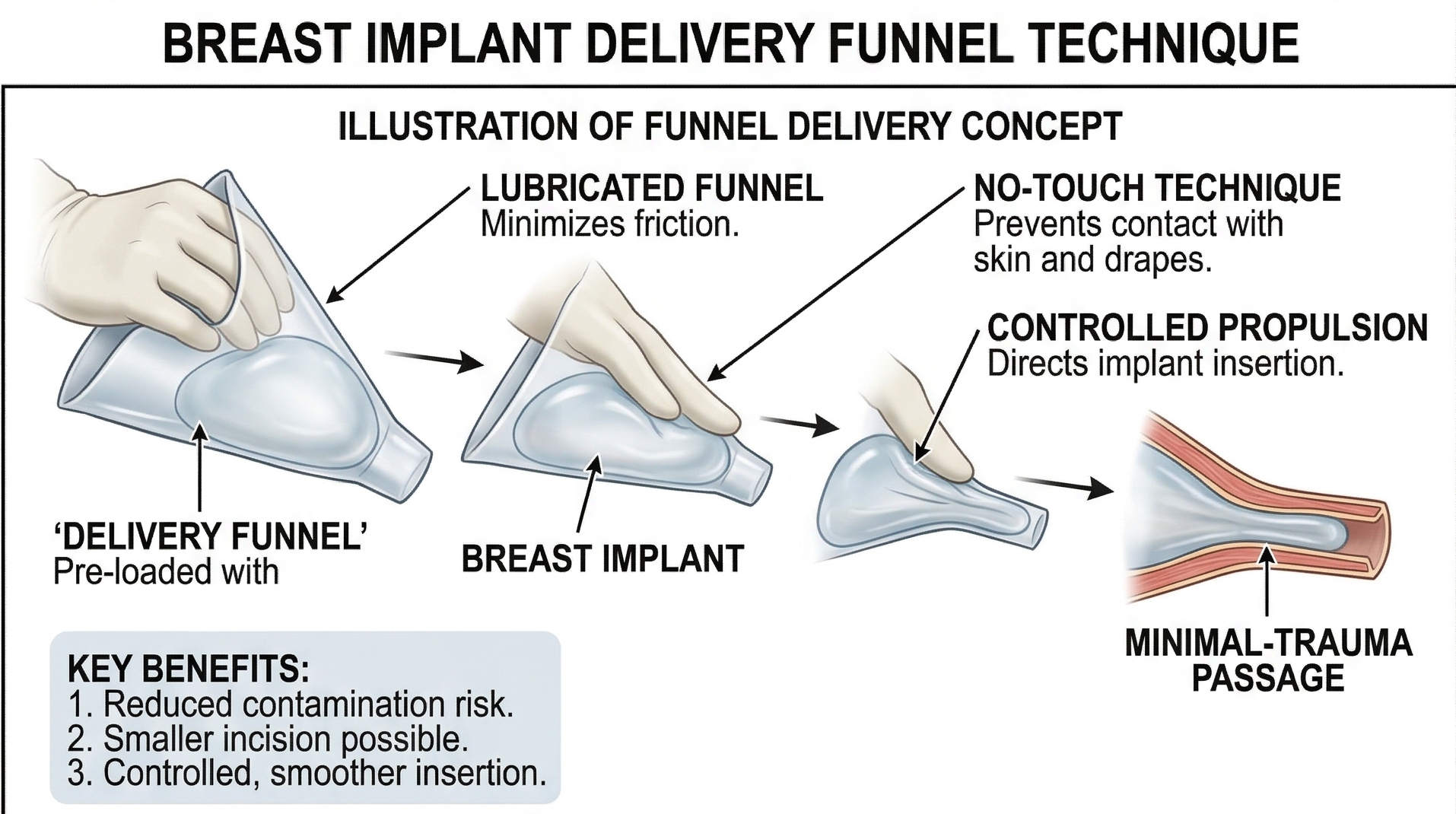
A cone-shaped delivery funnel slides the implant directly into the pocket without touching your skin at all.
Combined with a 14-point safety protocol, the no-touch approach drops capsular contracture rates to under 1% in published studies.
I use this technique for every breast augmentation I perform at my San Francisco practice, and I consider it non-negotiable.
1. What the No-Touch Technique Means for Your Breast Augmentation
The name is straightforward. During a no-touch breast augmentation, your implant never contacts your skin, the tissue lining the incision, or the breast gland itself as it travels into the surgical pocket. The implant goes from its sterile packaging, through a protective delivery sleeve, and directly into the space I have prepared for it, all without touching any surface carrying bacteria.
This matters because your skin is home to millions of bacteria. They live on the surface, inside hair follicles, and deep within the milk ducts of the breast. None of these organisms are dangerous under normal circumstances. But when they hitch a ride on a silicone implant and get sealed inside a closed surgical pocket, they do not go away on their own. They set up permanent colonies, and those colonies drive the most common complication in breast implant surgery: capsular contracture.
Dr. Richard Mladick, a Virginia-based plastic surgeon, formalized the no-touch concept for breast augmentation in a 1993 paper published in Aesthetic Plastic Surgery. He drew on aseptic principles dating back to the 1890s from orthopedic surgery, where keeping metal hardware free of skin bacteria was already recognized as critical.9 What Mladick did was apply those principles specifically to silicone and saline implants, proving the connection between contamination and capsular contracture with hard data from over 2,800 implants.
2. Why Implant Contamination Leads to Capsular Contracture
Every time your body encounters a foreign object, whether it is a splinter or a breast implant, the immune system surrounds it with a thin layer of scar tissue. In a normal breast augmentation recovery, this capsule is paper-thin, soft, and completely undetectable. You never know it is there.
Problems start when bacteria colonize the implant surface during insertion. The organisms most commonly responsible are Staphylococcus epidermidis, a species living on everyone's skin, and bacteria found inside the breast ducts. Once attached to the silicone shell, these bacteria produce a slimy protective layer called a biofilm. Within the biofilm, the bacteria slow their metabolism to a near standstill, making them invisible to your immune system and resistant to antibiotics.
Your body senses something is wrong but cannot clear the infection. The immune response ramps up chronically, releasing inflammatory signals instructing fibroblasts (the cells responsible for scar production) to keep laying down collagen around the implant. Over months and years, the capsule thickens, tightens, and contracts. The breast feels firm, looks distorted, and sometimes becomes painful. That is capsular contracture, and it is the single most common reason patients come to see me for revision surgery.
Here is the part most patients find surprising: the silicone shell itself makes contamination worse. Implant shells carry an electrostatic surface charge, similar to a balloon rubbed on a sweater, attracting microscopic debris the moment the device touches anything. Under electron microscopy, even smooth implants have microscopic craters and surface irregularities where bacteria tuck themselves in and start building biofilms. This is why preventing contact in the first place is so much more effective than trying to clean the implant afterward.
3. Capsular Contracture Rates Before and After the No-Touch Technique
The clinical data supporting the no-touch technique is some of the strongest in all of plastic surgery. Before Mladick published his protocol, capsular contracture rates across the field routinely ran between 10% and 20%.6 Many plastic surgeons at the time considered some degree of hardening almost inevitable, an unfortunate cost of having implants.
Mladick proved otherwise. In patients treated with his no-touch protocol between 1987 and 1992, the capsular contracture rate dropped to 0.6%, and the infection rate was zero. In a follow-up cohort of 2,000 implants in 1,000 patients, he reported only a single significant contracture.
More recently, Dr. William Adams tracked 4,176 implants over 20 years using a comprehensive 14-point safety bundle built on the no-touch foundation. His capsular contracture rate was 0.57%.41 A separate analysis of over 42,000 implants followed for an average of nearly 12 years showed a 2.2% contracture rate with strict protocol adherence, and zero cases of BIA-ALCL.
Those numbers are dramatic compared to historical averages. For patients in the Marina, Pacific Heights, or anywhere else across the Bay Area considering breast augmentation, I want you to understand: capsular contracture is not a lottery. It is largely preventable when your plastic surgeon follows the right protocol.
4. How the Delivery Funnel Changed the Game
Mladick's original technique relied on wide surgical retractors to hold the incision open while he passed the implant through without touching the skin. It worked, but it required extra assistants and significant force on the wound edges. The real turning point for widespread adoption came in 2009 when plastic surgeon Dr. Kevin Keller introduced a purpose-built implant delivery funnel, which received FDA Class I device approval.
The delivery funnel looks like a cone-shaped pastry bag made from transparent nylon. The inside is coated with a slippery hydrophilic material. Before insertion, I soak the funnel in sterile saline, which activates the coating and creates an almost frictionless interior surface. The implant drops into the wide end of the funnel straight from its sterile packaging. I trim the narrow end to match the implant size, slide the tip into the surgical pocket past all the skin and tissue edges, and then gently squeeze. The implant glides directly into the pocket like toothpaste from a tube.
The clinical benefits are well-documented:
A systematic review found funnel-assisted delivery reduced capsular contracture risk with a relative risk of 0.42 (meaning a 58% lower likelihood of developing contracture compared to traditional insertion).
One study reported a 10% contracture rate with hand insertion versus 1.3% with the funnel.
The average insertion time drops to about 6 seconds, limiting how long the implant is exposed to the operating room air.
The funnel allows larger cohesive gel implants to pass through smaller incisions, typically about 35mm, reducing visible scarring.
I tell my patients from Tendernob and SoMa to think of it this way: the delivery sleeve is a sterile tunnel between the packaging and your body. The implant goes in clean and stays clean.
5. The 14-Point Plan: Where the No-Touch Technique Fits into a Complete Safety Protocol
The no-touch insertion is one piece of a larger puzzle. Dr. William Adams and Dr. Anand Deva developed the 14-Point Plan around 2013-2014, a standardized surgical checklist designed to prevent biofilm formation, capsular contracture, and BIA-ALCL (a rare lymphoma linked to chronic implant-related inflammation). The no-touch delivery step sits at the center of this plan, but every other step matters too.
The key elements I follow for every breast augmentation:
Intravenous antibiotics at the time of anesthesia induction, not after the case starts. This gives the medication time to reach effective tissue levels before I make the first incision.
Inframammary fold incision. The periareolar approach cuts through the milk ducts, which harbor their own bacterial population. Studies show periareolar incisions carry a 7.2% capsular contracture rate versus 3.1% for the inframammary approach.
Nipple shields placed over both areolae to prevent duct bacteria from seeping onto the surgical field during tissue handling.
Minimal tissue trauma during pocket dissection. Blood and bruised tissue act as a nutrient-rich culture medium for any bacteria present.
Triple-antibiotic pocket irrigation after dissection and before implant delivery, washing away any planktonic bacteria introduced during pocket creation.
Complete glove change before touching the implant or the funnel. The gloves I wore during dissection have contacted skin, fat, and tissue. Fresh gloves mean the implant is handled by a pristine surface.
Funnel-assisted no-touch delivery. The implant passes from sterile packaging through the funnel and into the pocket without contacting any biological surface.
This protocol is not complicated, but it demands discipline at every step. Each individual measure contributes a layer of protection, and together they reduce contamination risk far below what any single step accomplishes alone.
6. The Connection Between Implant Contamination and BIA-ALCL
I bring up BIA-ALCL (breast implant-associated anaplastic large cell lymphoma) not to alarm you, but because it is the most serious reason the no-touch technique matters. BIA-ALCL is a rare T-cell lymphoma occurring in the capsule or fluid around breast implants, most commonly associated with heavily textured implant surfaces.
The leading hypothesis points to chronic, low-grade bacterial inflammation as a triggering factor. Researchers have identified specific organisms, including Ralstonia pickettii, a water-borne bacteria, in the capsules of BIA-ALCL patients.2 The theory is straightforward: persistent biofilm drives persistent inflammation, and in genetically susceptible individuals, years of unresolved inflammation gives rise to malignant cell transformation.
In the large-scale study of over 42,000 implants tracked for nearly 12 years with strict 14-point plan adherence, the BIA-ALCL rate was zero. Zero cases. For patients in the East Bay and South San Francisco thinking about breast augmentation, I want to make the point clearly: meticulous sterile technique during your surgery has implications far beyond how soft your breasts feel. It is an oncological precaution.
7. How I Apply the No-Touch Technique at My San Francisco Practice
I have been performing breast augmentation for over 25 years, and the no-touch technique has been part of my standard protocol for the majority of my career. Here is how the insertion portion of a typical augmentation goes in my operating room on Sutter Street:
After dissecting the pocket and completing hemostasis, I irrigate aggressively with an antibiotic solution. Every instrument used during dissection goes onto a separate tray and does not come back to the surgical field. My scrub tech opens a fresh pair of gloves, and I re-gown my hands completely. The implant packaging is opened, and the device goes straight into a pre-soaked delivery funnel without being placed on any surface. The narrow end of the funnel enters the incision past the skin edges, and the implant is delivered into the pocket with gentle compression. Total skin contact: zero.
For the second breast, we repeat the entire protocol with a new funnel. This is not optional. Using the same funnel for both sides creates a documented cross-contamination risk. Two funnels, two sets of gloves, two separate rounds of irrigation. The extra time and cost are trivial compared to the revision surgery needed to address a capsular contracture.
I pair the no-touch approach with subfascial or dual-plane implant placement and my Rapid Recovery protocol, so patients from Marin to San Mateo head home the same day and return to their normal routines faster than they expected. The no-touch insertion is not something you see or feel as a patient. But it is the invisible foundation keeping your results stable for years to come.
Myths About the No-Touch Technique
Myth: Capsular contracture is random and unavoidable. Decades of clinical data say otherwise. When Mladick isolated the implant from skin bacteria, contracture rates fell from double digits to under 1%.6 Contamination during insertion is the primary driver, and contamination is preventable.
Myth: The delivery funnel is a marketing gimmick. The funnel received FDA device approval in 2009 and has been validated in systematic reviews and multi-center studies.3 A relative risk reduction of 0.42 for capsular contracture is not a gimmick. It is a statistically significant clinical improvement.
Myth: Antibiotics in the pocket solve the contamination problem. Pocket irrigation is an important step, but it only works against free-floating bacteria. Once organisms attach to the silicone surface and form a biofilm, standard antibiotics are largely ineffective.4 The no-touch technique prevents bacteria from reaching the implant in the first place, which is a fundamentally different strategy than trying to kill them after the fact.
Myth: The no-touch technique only applies to silicone gel implants. Mladick developed his protocol using saline implants in the early 1990s, years before silicone gel returned to the market. The principle applies to any breast implant because the electrostatic surface charge attracting contaminants is a property of the silicone elastomer shell, and both saline and gel implants share that same shell material.
Frequently Asked Questions
-
Barely. With a delivery funnel, the implant insertion itself takes about 6 seconds. Changing gloves and opening a second funnel for the other side adds a few minutes to the overall case. The time difference is negligible, and the benefit in reduced complication risk is significant.
-
The delivery funnel and the additional sterile supplies add a small cost per case, but at my practice this is built into the surgical fee. I do not charge separately for following what I consider baseline safety measures. If a plastic surgeon is not using some form of no-touch delivery at this point, I would ask why.
-
Plastic surgeons grade capsular contracture on a four-level scale. Grade I means the breast looks and feels normal. Grade II feels slightly firm but looks fine. Grade III is visibly distorted with noticeable firmness. Grade IV adds pain to the distortion. Grades III and IV typically require surgical revision.
-
BIA-ALCL is a rare lymphoma linked to chronic bacterial inflammation around implants. The leading research hypothesis is that biofilm-driven inflammation, sustained over years, triggers malignant cell changes in susceptible patients. By preventing the bacterial contamination responsible for biofilm formation, the no-touch technique addresses the root cause identified in the BIA-ALCL pathway. In a study following over 42,000 implants with strict protocol adherence, zero cases of BIA-ALCL were reported.
-
Not all of them, and protocols vary from practice to practice. The 14-point plan is considered the global standard, but adoption is not universal. During your consultation, ask your plastic surgeon specifically how the implant will be handled during insertion. Ask about glove changes, funnel use, and antibiotic irrigation. A confident, experienced plastic surgeon should be happy to walk you through their protocol.
-
The cone-shaped delivery funnel is the most widely studied device, but other validated approaches exist. Some plastic surgeons use light-handle covers or reversed surgical glove sleeves built on the same principle: creating a sterile, low-friction barrier between the implant and the skin. The specific device matters less than the commitment to no-touch principles. What you want to avoid is any technique where the implant is pushed through the incision by hand with direct skin contact.
Summary
The no-touch technique addresses the single biggest risk factor in breast augmentation: bacterial contamination of the implant during insertion. By keeping the device completely isolated from skin and tissue bacteria using a delivery funnel and strict sterile protocol, your plastic surgeon eliminates the primary trigger for capsular contracture and reduces the theoretical risk of BIA-ALCL. This is not new or experimental. The concept has been validated since 1993, refined through modern delivery funnels and the 14-Point Plan, and backed by data from tens of thousands of patients over decades. I consider it the minimum standard for any breast augmentation in 2026.
Schedule Your Consultation
If you are considering breast augmentation and want to know exactly how your implant will be handled during surgery, I am happy to walk you through my protocol in detail during a one-on-one consultation at my San Francisco office at 450 Sutter Street, Suite 1440. Call 415-362-1846 or reach out through my website to set up an appointment. I also see patients at my Alameda office at 1403 Park Street for those coming from the East Bay.
Follow and Connect with me on my social media!